The Glasgow Coma Scale provides a clinical index of the ‘overall’ acute impairment of brain function, or so called ‘conscious level’. When it was described, features of focal dysfunction such as brainstem signs were not incorporated and instead were intended to be assessed separately. Nevertheless, there have been views that indices or scores that combine an assessment of ‘conscious level’ and ‘brain stem’ function may have a useful place.1-3 In response, the GCS Pupils Score (GCS-P) was constructed to bring together information about a patient’s responsiveness as reflected in the GCS Score and the pupil reaction4(Link).
The GCS Score is derived by assigning notation to the level in each of the three subcomponents of the GCS and summing the results of a patient’s assessment. Further information on the GCS Score can be accessed here. The Pupil Reactivity Score summarises Information about loss of pupil reactivity to light and is calculated as follows.
| Pupils Unreactive to Light | Pupil Reactivity Score |
|---|---|
| Both Pupils | 2 |
| One Pupil | 1 |
| Neither Pupil | 0 |
The GCS-P is calculated by subtracting the Pupil Reactivity Score (PRS) from the Glasgow Coma Scale (GCS) total score:
GCS-P = GCS - PRS
Relation to outcome
The relationship between the combination of GCS Score and pupil score, and severity of a head injury as reflected in outcome, was examined in information on 15,900 patients drawn from the two largest available data sets (IMPACT and CRASH).
Impairment of pupil reactivity was associated with a worsening of outcome across the range of GCS Scores. There was a continuous, fundamentally smooth, relationship between the combined GCS –P Score and mortality or failure to achieve an independent outcome.
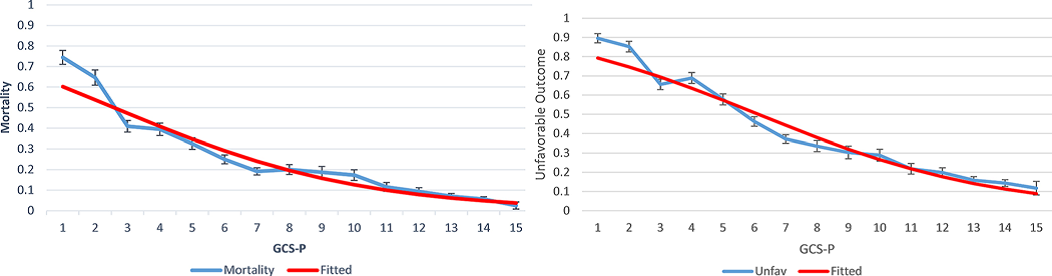
Figure 1. Mortality 6 months after head injury (left) and unfavourable outcomes (right) according to patient GCS-P score at admission. Error bars show 95% confidence intervals. The fitted lines come from logistic regression models in which the GCS-P was treated as a continuous variable.4 Examination of the information yield measured using Nagelkerke’s R2 confirmed that the combination was superior to either clinical feature alone and that the information from the simple arithmetic score was very close to that calculated by more complex models for integration
Role of GCS-P Score
The combined GCS-P is not intended to replace the role of separate assessment and reporting of each component of the Glasgow Coma Scale and pupil response in the care of individual patients. It expands on the GCS Score as a simple shorthand index of the severity of a patient’s clinical state and prognosis, especially in more severe injuries. It may be useful in avoiding bias in decisions from individual clinicians’ perceptions, in drawing up guidelines and in describing findings in groups of patients.5 The GCS-P provides a uni-dimensional index of clinical severity onto which information about other key prognostic features, such as age, can be added in a simple format likely to be useful in clinical practice6 (GCS-P Charts).
References
- Benzer A, Mitterschiffthaler G, Marosi M, Luef G, Pühringer F, De La Renotiere K, et al: Prediction of non-survival after trauma: Innsbruck Coma Scale. Lancet 338:977–978, 1991
- Born JD: The Glasgow-Liège Scale. Prognostic value and evolution of motor response and brain stem reflexes after severe head injury. Acta Neurochir (Wien) 91:1–11, 1988
- 3. Wijdicks EFM, Bamlet WR, Maramattom BV, Manno EM, McClelland RL: Validation of a new coma scale: The FOUR score. Ann Neurol 58:585–593, 2005
- Brennan PM, Murray GD, Teasdale GM. Simplifying the use of prognostic information in traumatic brain injury. Part 1: The GCS-Pupils score: an extended index of clinical severity J Neurosurg 128:1612–1620, 2018
- Moore NA, Brennan PM, Baillie JK: Wide variation and systematic bias in expert clinicians’ perceptions of prognosis following brain injury. Br J Neurosurg 27:340–343, 2013
- Murray GD, Brennan PM, Teasdale GM: Simplifying the use of prognostic information in traumatic brain injury. Part 2: Graphical presentation of probabilities. J Neurosurg. 128(6):1621-1634. 2018
Practical Example
You are asked to assess a seriously head injured patient. They make no eye, verbal or motor movements, either spontaneously or in response to your spoken requests; when stimulated with fingertip pressure their eyes do not open, they make no sounds, but their arms flex normally. This translates to scores of E1V1M4 giving a sum score of 6 which carries a likelihood of death of 29%. Neither pupil is reactive to light; this gives a Pupil Reactivity Score (PRS) of 2. The GCS-P score (GCS-PRS ) is 6-2 = 4. The likelihood of mortality is increased to 39%.

